
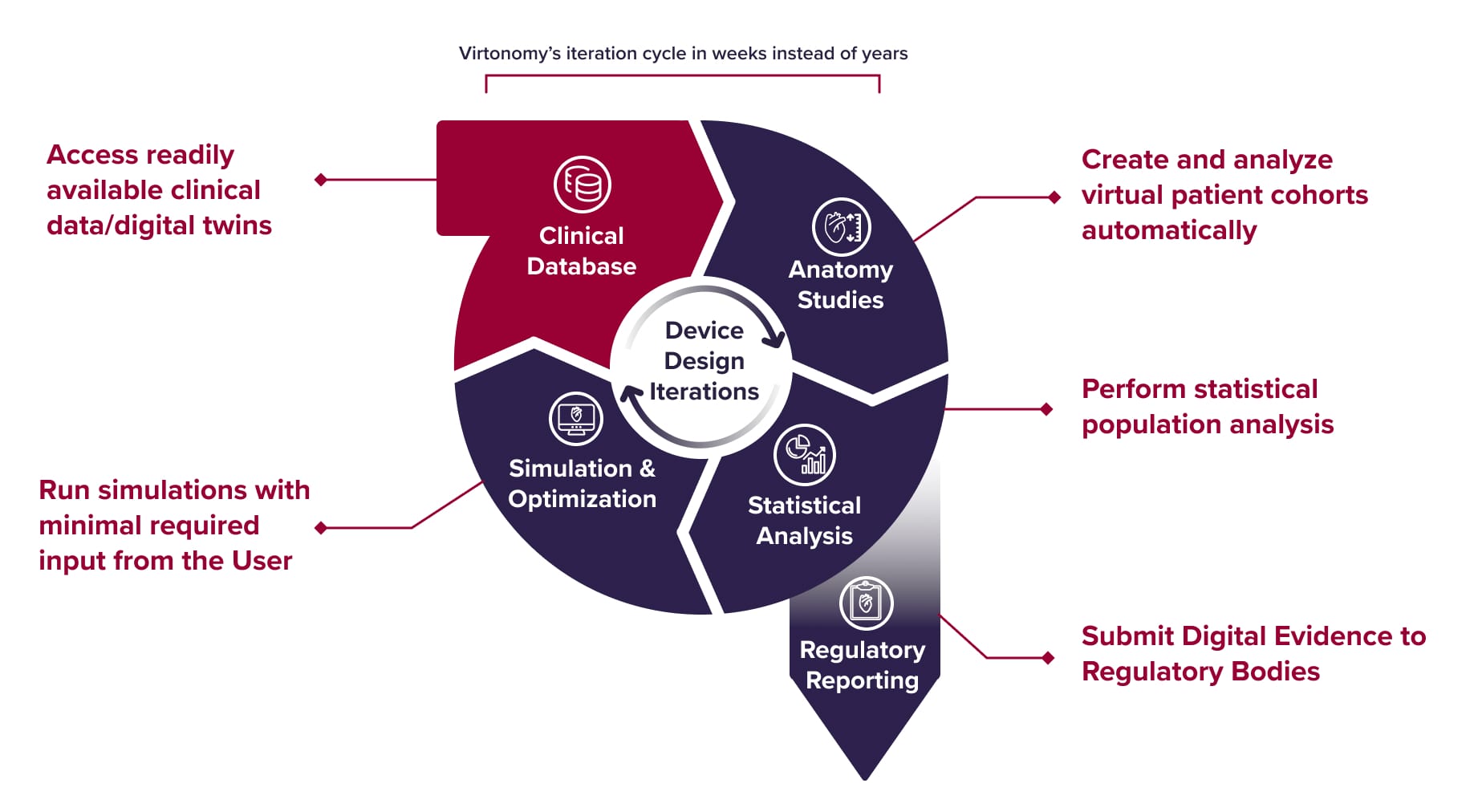
How it works
Virtonomy’s v-Patients software allows medical device developers to conduct development and testing within a virtual environment. The solution leverages a comprehensive digital twin and simulation platform built on a continually growing database of real clinical data, accurately representing anatomical differences, demographic diversity, and various pathological conditions.

Computer simulations
Digital patient twins
Statistical analysis
Regulatory reports
Our end-to-end digital twin solution
Virtonomy’s virtual trial platform supports the full product life cycle of medical device development, from concept phase to regulatory reporting and clinical practice
Thousands of Digital Patient Twins
Select your virtual patient population from our extensive database of humans and animals based on real CT scans. Choose based on disease, age, sex, ethnicity, ejection fraction, and your anatomical parameters. Perform virtual implant fitting, interactive measurements, and collision detection.
3D measurements ensure high accuracy in contrast to measuring on 2D CT scan slices. Our expertise allows for complex measurements, including cardiovascular strain and loading conditions analysis.
Main features:
- Virtual patient cohort tailored for you
- Advanced 3D visualization and interactive implantation
- Accurate & reproducable 3D measurements and extensive statistics


Advanced Statistical Shape Models
Select your chosen patient group and create the average patient anatomy automatically. Investigate different age groups, ethnicities, and only female or male patients. Combine the anatomical variations and investigate their influence on your design.
Identify worst-case anatomies and improve your device coverage. Extend the indications of use to treat the maximum number of patients resulting in major financial benefits.
Main features:
- Worst and edge-case anatomies
- Average patient anatomy
- Extensive statistics of demographics & measurements
Advanced Anatomical Simulations
Simulate your implant or product family in the entire virtual target population to investigate how these body variations may affect the performance and safety of your device. In addition, identify the potential for design optimization and material selection by conducting parameter analyses.
We offer a device and use case tailored customized simulation environment based on your use case and product phase. Get access to an easy to use, intuitive solution where no expert simulation knowledge is needed.
Main features:
- You control the simulations, we take care of all difficulties
- Validated in-human implant simulations
- Validated ISO benchtop test simulations
ISO 5840:2021 Cardiovascular implants — Cardiac valve prostheses
ISO 5910:2018 Cardiovascular implants and extracorporeal systems — Cardiac valve repair devices
ISO 7198:2016 Cardiovascular implants and extracorporeal systems — Vascular prostheses — Tubular vascular grafts and vascular patches
ISO 18193:2021 Cardiovascular implants and artificial organs — Cannulae for extracorporeal circulation
ISO 18242:2016 Cardiovascular implants and extracorporeal systems — Centrifugal blood pumps
ISO 22679:2021 Cardiovascular implants — Transcatheter cardiac occluders
ISO 25539:2017 Cardiovascular implants — Endovascular devices
Other benchtop tests are available upon request


Regulatory Services & Consultation
Compared to conventional in vivo and in vitro testing, in silico trials of implants are more time and cost efficient. We provide digital evidence of your anatomy, statistical and simulation results to support your technical documentation with FDA-compliant reporting (FDA 1807 & ASME V&V40).
Ask our regulatory experts to establish your virtual testing plan connected to your physical testing. Learn about the new FDA guidance documents and ASME standards for Computational Modeling and Simulation. Our experts will highlight how you can apply them and streamline your regulatory process.
Main features:
- Regulatory path with a virtual testing plan
- Compliance with all regulatory bodies
- Validated virtual evidence report
Watch an introductory video of the product and its possibilities
Check the video to see how to set up your project in our product v-Patients, and to see what it can be used for.

OUR DATABASE
We develop our digital twins from real imaging data.
You can access our database of thousands of human and animal 3D models segmented from real CT and MRI scans. Whole heart and full thorax models are available including every organ.
Physiological data brings the patients “alive” and enable anatomical simulations. Tissue material models (elasticity parameters), blood flow properties (pressure & velocity) and electrocardiogram (ECG) data expand our anatomical database.



The data and the analysis that Virtonomy has provided during our partnership are key tools for strengthening and expanding our knowledge basis of the anatomical boundary conditions.

Davide Mogentale, R&D Manager
Medira GmbH
OUR SIMULATION FEATURE
Access high-level simulations via an intuitive interface


Advanced, automatic simulations for many patients – in 2 steps

Access validated simulation scenarios readily available
We integrate your device into your special simulation scenario.
Validated simulations

Run simulations for many patients through an intuitive interface
Ensure maximum population coverage by testing your device on your patient cohort
1000+ virtual patients
Only tune what you want to investigate – we take care of the rest
- Choose patients and their pathology for the simulation
- Choose boundary conditions to investigate their influence
- Access a library of material models for your device
- Define automated reports generated for each simulation


We ensure a smooth user experience
- Seamless cloud computation, no need for local workstations
- Accurate time estimation when starting a simulation
- Fast device positioning for new simulations
- Automatic setup of numerical parameters, no mistakes
Virtonomy supports you from initial design idea to regulatory submission
v-Patients: From clinical data to digital evidence
Exemplary use case of a transcatheter valve replacement

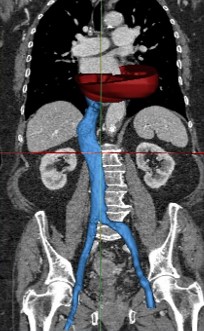
Huge Database of Virtual Patients
Analyse the right heart and transfemoral access
route for the implantation of a transcatheter valve
application
Automatic pipeline from data to model using in-house developed AI & Computer Vision

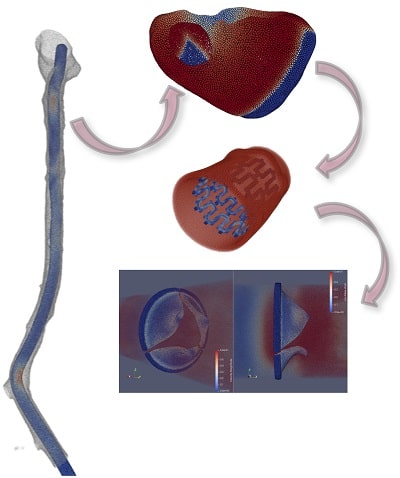
Worst- and average-case assessment
Perform statistical analysis of 50+ right heart patients to define inclusion/exclusion and sizing criteria, assess worst case scenarios, and more
Huge database in the cardiovascular field allows us to perform high diversity analysis

Advanced medical simulations
Perform solid, fluid and electrophysiology simulations to predict device tissue-blood interaction and evaluate strain, stress, and more
Developing the most advanced browser-based simulation framework for healthcare applications

Digital evidence for regulatory submission
Create regulatory-compliant digital evidence of your results to support your technical documentation or get support in drafting a testing plan from our team of experts
Automatic regulators-compliant report revolutionizing and accelerating the submission process

Also available in VR for an immersive experience!
Use the virtual reality mode and view anatomy and different anatomical measurements, investigate device fit through a virtual device implantation and perform simulations.



Virtual implantation brings enormous benefits to speeding up product development in a data driven manner and complementing the animal studies. Now VR technology just takes this to the next new level, and it is exciting to develop an artificial organ in the metaverse!

Oliver Chu, Principal Engineer
Scandinavian RealHeart

Ready to cut your device development time and costs?
We are passionate about helping medical device developers shorten their time-to-market and develop new products faster.

