The medical device industry has long measured progress in years saved, lives improved, and regulatory hurdles cleared. But there is another metric that rarely appears on development dashboards: the carbon footprint of the testing process itself.
When we talk about making healthcare more sustainable, the conversation usually focuses on hospitals, packaging, or supply chains. Rarely does it touch on something that happens much earlier in the process: the animal experiments used to test medical devices before they ever reach a patient.
Testing a new heart valve or stent on a living animal, usually a pig, calf or sheep, has been the standard for decades. But it comes with a carbon footprint that the industry has largely ignored. Digital twins and computer simulations offer a cleaner alternative. The difference in environmental impact is bigger than most people would expect.
And the timing matters. In 2025, the FDA announced plans to phase out mandatory animal testing for many drug and device development programmes — officially recognising in silico simulation as a valid alternative for regulatory submissions. The science, the regulation, and the sustainability case are all pointing in the same direction.
The True Carbon Cost of One Animal Experiment
To understand what is at stake, it helps to walk through the full lifecycle of a single preclinical animal study. Every stage carries an emissions burden.
Breeding the animal
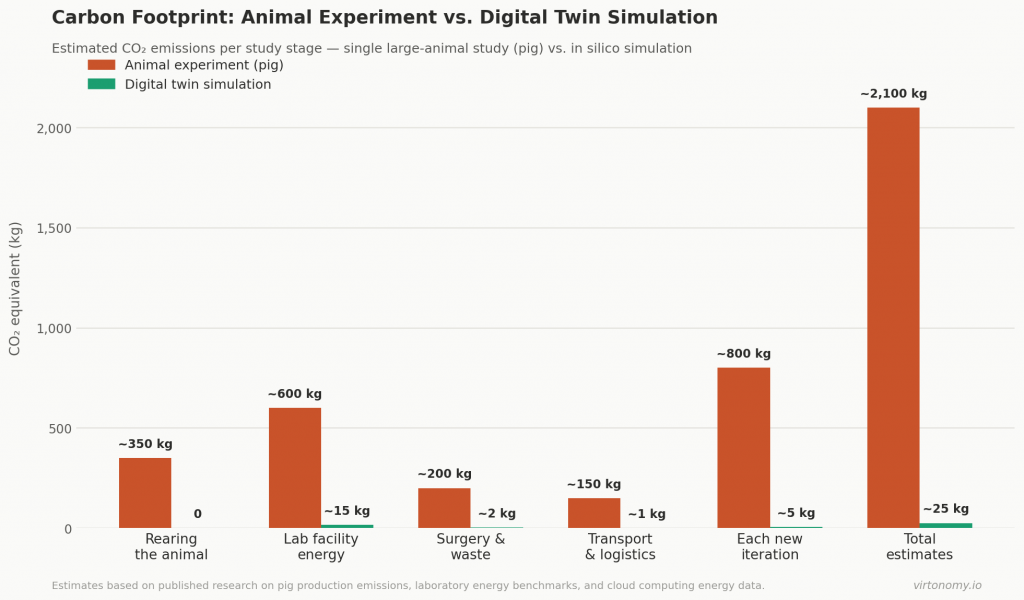
A large animal like a pig used in cardiovascular research typically weighs 60–80 kg at the time of the experiment. Research on pig production systems shows that the carbon footprint of pork production ranges from approximately 3 to 7 kg CO₂-equivalent per kilogram of live weight — meaning just rearing one preclinical pig can represent 200–500 kg CO₂ before the study even begins. Feed production alone — growing, processing, and transporting feed crops — accounts for the largest share of this figure.
The laboratory facility
Animal research facilities consume a lot of energy. The constant ventilation, heating, cooling, and waste management systems that keep lab animals alive and conditions controlled run continuously — 24 hours a day, seven days a week, for the entire duration of the study.
The experiment itself
Surgical procedures, anaesthesia delivery, post-operative monitoring, consumables (sterile drapes, tubing, imaging contrast agents, single-use instruments), and veterinary staff all generate emissions and waste. Each year, research laboratories collectively produce an estimated 5.5 million metric tons of plastic waste globally.
Transport and logistics
Animals must be transported from breeding facilities to research sites. Equipment, reagents, and implants must be shipped. In multi-centre studies, human teams travel between locations.
Repetition
But the biggest multiplier of all? Most experiments don’t work the first time. Device development is iterative — a design fails, engineers tweak it, and the whole cycle begins again. Every new iteration means starting from scratch: a new animal, a new facility booking, a new round of logistics.
Add it all up, and a single animal study can generate somewhere between 1,000 and 3,000 kg of CO₂. Run a study with ten animals across two or three rounds of testing, and you are looking at 20–30 tonnes of CO₂ — roughly the same as two or three years of an average European citizen’s entire carbon footprint.
What does a Digital Twin Study Emit instead?
Now consider the alternative: a computational simulation study using virtual patient cohorts, such as those generated by Virtonomy’s v-Patients platform.
The primary energy cost of a digital twin study is server compute time. A week-long 3D simulation does draw real power. But there are easy options to revert these drawbacks.
Data centres running on renewable energy
Major cloud computing providers have made substantial investments in renewable energy sources. When simulation workloads are run on green-powered infrastructure, the marginal emissions per simulation hour are a fraction of what the traditional testing methods consume — and often close to zero on a net basis.
No biological supply chain
There is no animal to breed, feed, house, transport, or care for post-operatively. There are no single-use surgical consumables, no anaesthetic agents to produce and ship, no biological waste streams to manage.
Instant iteration
When a device design changes, updating a virtual model and re-running a simulation takes hours to days, not weeks or months. There is no new animal to procure, no facility slot to book, no ethical committee re-approval required. Each iteration adds marginal compute cost, not an entirely new experimental lifecycle.
Scale without proportional cost
A single v-Patients study can simultaneously test a device across hundreds of virtual patient anatomies — capturing variability in sex, age, body size, and pathology that would be practically and ethically impossible to replicate in live animal cohorts of comparable size. The compute cost of testing across 200 virtual patients is not 200 times greater than testing on one.
Evidence from the manufacturing sector illustrates the scale of the opportunity. Research presented at MedtecLIVE demonstrated that using digital twin technology and finite element analysis in the development of medical components — specifically for titanium deep-drawn parts — reduced the carbon footprint during the development phase by up to 70% compared to conventional physical prototyping methods. Even accounting for the energy consumed by week-long high-performance computing simulations, the environmental savings were substantial and measurable.
| One Animal Study (pig) | One Digital Twin Study | |
|---|---|---|
| Raising the animal | ~200–500 kg CO₂ | None |
| Facility energy | Very high (runs 24/7) | Low (cloud computing) |
| Surgical waste & consumables | Significant | Minimal |
| Transport | Moderate to high | Near zero |
| Cost of each new iteration | Full restart | Small compute cost |
| Patients tested | Typically 1–10 animals | Hundreds of virtual patients |
| Total CO₂ estimate | 1,000–3,000+ kg | Tens of kg |
The gap is not small. And regulators are catching up — the FDA and other agencies are increasingly accepting simulation-based evidence as part of medical device approvals, meaning that choosing the lower-carbon path does not mean compromising on scientific standards.
Conclusion
People sometimes assume that being more sustainable in medical research means slowing things down or cutting corners. The numbers above suggest the opposite. Switching from animal experiments to digital simulations is faster, covers more patient types, and produces a fraction of the carbon emissions.
At Virtonomy, our v-Patients platform lets device developers run their preclinical testing virtually, using patient models built from real clinical data. The result is evidence that regulators accept, development that moves faster, and a significantly smaller environmental footprint — without a single animal involved.
The technology to make this shift exists today. The question is simply how quickly the industry chooses to use it. And with FDA planning to scrap animal testing, medical device companies should look for alternatives fast.




